Loading...
Loading...
Sterile injectable formulations including vials, ampoules, and pre-filled syringes manufactured in certified aseptic facilities.

Good Manufacturing Practice
Quality Management
World Health Organization
Drug Controller General

Vials
Amikacin-100mg Inj

Vials
Amikacin-250mg Inj

Vials
Amikacin-500mg Inj

Vials
Cefoperazone 1000mg+Sulbactam 500gm
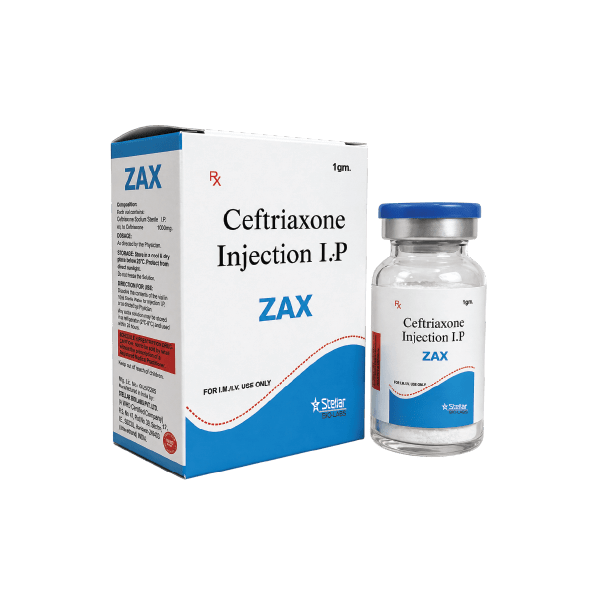
Vials
Ceftriaxone 1000 mg
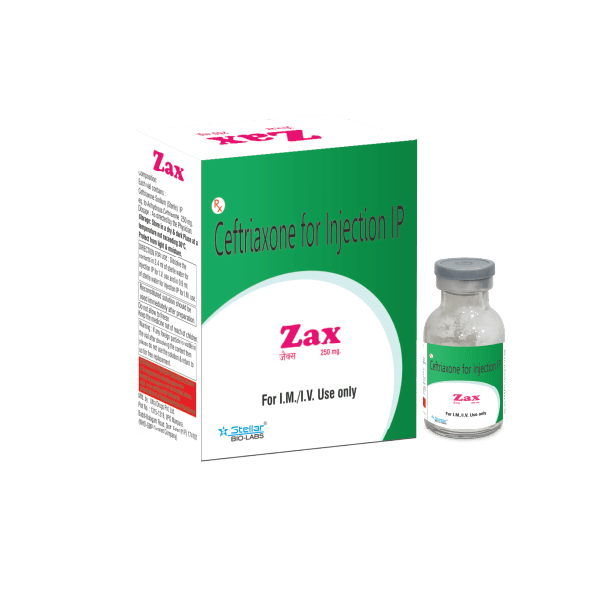
Vials
Ceftriaxone 250 mg
Aseptically manufactured sterile formulations
Dedicated sterile manufacturing area with Class 100 laminar air flow and real-time environmental monitoring.
Every batch undergoes LAL endotoxin testing and sterility verification per pharmacopeial standards.
Injectable formulations command premium pricing with strong hospital and institutional demand.
Our Injectables Range is manufactured in a state-of-the-art aseptic facility with Class 100 laminar airflow systems, HEPA filtration, and continuous environmental monitoring. From reconstitutable dry powder vials to ready-to-use ampoules, every product meets stringent sterility and pyrogen-free standards.
“We are committed to delivering pharmaceutical excellence through rigorous quality control, cutting-edge research, and unwavering dedication to healthcare innovation.”
"The quality is consistently excellent. WHO-GMP certification gives confidence. Patients trust the brand."
"Best support system. From training to marketing, everything provided. Team treats you like family."
"Timely delivery and excellent packaging. Never had availability issues. Great experience over 3 years."
"Professional approach and transparent dealings. Regular visits by support team ensure smooth operations."
"Excellent product range and competitive pricing. Market support materials help in doctor visits."
"Monopoly rights are genuine. No competition from same company. Territory protection is real advantage."
Contact us for inquiries, partnerships, or product information
We'll respond within 24 hours